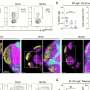
Research conducted by scientists at the University of Manchester has provided significant insights into how the immune system in the gut changes after a stroke and the potential implications for gastrointestinal health. The study, published in the journal Brain, Behavior and Immunity, explores the concept of the “gut-brain axis,” which posits that communication exists between the gut and the brain, influencing both physical health and disease states.
Stroke, a critical medical emergency that disrupts blood flow to the brain, often results in severe long-term effects on a patient’s mobility and cognitive functions. Patients recovering from a stroke frequently face secondary complications, including bacterial infections and gastrointestinal issues such as swallowing difficulties and constipation. The research highlights a growing body of evidence that suggests these complications are linked to alterations in the gut microbiota—the community of beneficial bacteria essential for gut health.
While previous studies have indicated changes in gut microbiota in stroke patients and animal models, the reasons behind these alterations and their significance for stroke recovery have been inadequately understood. The latest findings reveal that signals from the nervous system may influence gut immune responses after a stroke.
New Discoveries on Immune Response
The research team investigated what occurs in the small intestine following a stroke in mice, discovering that populations of immune cells responsible for antibody production changed significantly within days of the event. Notably, a specialized group of cells that produce an antibody known as Immunoglobulin A (IgA) became hyper-activated. IgA plays a crucial role in regulating the populations of commensal bacteria within the intestine, thereby influencing overall gut health.
Remarkably, the study also found that mice lacking IgA did not display the same degree of changes in their gut microbiome after a stroke. This observation suggests that alterations in immune function could partially explain the gastrointestinal symptoms experienced by stroke patients.
Lead investigator, Professor Matt Hepworth from the Lydia Becker Institute of Immunity and Inflammation, emphasized the dual impact of stroke, stating, “Stroke is a devastating neurological event that also has many long-term consequences, leaving patients at risk of airway infections and gastrointestinal complications.” He highlighted the collaboration with neuroscientists to uncover how immune disturbances in the gut may contribute to these complications.
The study not only enhances the understanding of stroke-related pathologies but also opens avenues for potential therapeutic interventions. “As immune-targeting therapeutics are increasingly used in the clinic, this research paves the way for addressing immune-driven disease symptoms following a stroke, which could significantly improve patients’ quality of life,” Professor Hepworth added.
Implications for Future Research
These findings point to the need for further investigation into the role of the gut-brain axis in stroke recovery and the potential for treatments that target immune system alterations. Understanding these interactions could lead to new strategies for managing the long-term complications associated with strokes and improving recovery outcomes for patients.
The comprehensive study, authored by Madeleine Hurry and colleagues, is set to be published in 2026 and could shape future research efforts in this vital area of health science. The insights gained from this work underscore the intricate relationship between the gut and brain, particularly in the context of neurological emergencies like stroke, and their lasting impact on patient health.